Ready to transform your health?
Unlock access to expert guidance and a weight care plan crafted just for you.
Similar Articles
Similar Articles

How Long Does Semaglutide Stay in Your System?
How Long Does Semaglutide Stay in Your System?

How to Spot Fake Semaglutide: Safety Risks and Warning Signs
How to Spot Fake Semaglutide: Safety Risks and Warning Signs

Alternatives to BMI: Better Ways to Measure Health
Alternatives to BMI: Better Ways to Measure Health
What Is Orforglipron? The New GLP-1 Weight Loss Pill Explained
What Is Orforglipron? The New GLP-1 Weight Loss Pill Explained
Is orforglipron right for you? Everything you need to know about Foundayo, the new oral GLP-1 pill for weight loss, from clinical results to cost.
Is orforglipron right for you? Everything you need to know about Foundayo, the new oral GLP-1 pill for weight loss, from clinical results to cost.

Table of Contents
Table of Contents
Key Takeaways
What Is Orforglipron and How Does It Work?
What Did the ATTAIN-1 Clinical Trial Show?
What Are the Side Effects of Orforglipron?
How Is Orforglipron Different From Other GLP-1 Medications?
How Much Does Orforglipron Cost?Who Is Eligible for Orforglipron?
FAQ'sCheck Your Eligibility
Key Takeaways
What Is Orforglipron and How Does It Work?
What Did the ATTAIN-1 Clinical Trial Show?
What Are the Side Effects of Orforglipron?
How Is Orforglipron Different From Other GLP-1 Medications?
How Much Does Orforglipron Cost?Who Is Eligible for Orforglipron?
FAQ'sCheck Your Eligibility
Key Takeaways
Orforglipron (brand name Foundayo) is a nonpeptide oral GLP-1 pill for weight loss, FDA-approved on April 1, 2026
Unlike injectable GLP-1 medications, orforglipron can be taken any time of day without food or water restrictions
In the ATTAIN-1 clinical trial, participants on 12 mg of orforglipron lost an average of 8.4% of their body weight over 72 weeks (the FDA-approved doses range up to 17.2 mg)
The most common side effects are gastrointestinal, including nausea, constipation, and diarrhea, and were mostly mild to moderate in clinical trials
Without insurance Foundayo will cost $149 per month at the lowest dose. At Mochi Health, you can also speak with an independent licensed provider about compounded orforglipron options, starting at $58.50 per month.
Orforglipron is a new FDA-approved oral weight loss medication, sold under the brand name Foundayo and manufactured by Eli Lilly (1, 3). It is a small molecule (non-peptide) GLP-1 receptor agonist which was approved on April 1, 2026, for use in adults with obesity or overweight with at least one weight-related condition (1). Unlike injectable GLP-1 receptor agonists, orforglipron is a once-daily oral medication that you can take at any time of day, with or without food, and without any water restrictions (3).
If you have been keeping up with the GLP-1 medications for weight loss, you will recognize how huge this is! In this article, we will cover: How orforglipron works, Results of the clinical trial, Side effects, How it compares to other GLP-1 receptor agonists, Cost of the medication, and Who may be eligable.
What Is Orforglipron and How Does It Work?
Orforglipron is a new FDA-approved oral weight loss medication, sold under the brand name Foundayo and manufactured by Eli Lilly (1, 3). It is a small molecule (non-peptide) GLP-1 receptor agonist which was approved on April 1, 2026, for use in adults with obesity or overweight with at least one weight-related condition (1). Unlike injectable GLP-1 receptor agonists like semaglutide or tirzepatide, orforglipron is a once-daily oral medication that you can take at any time of day, with or without food, and without any water restrictions (3).
GLP-1 (glucagon-like peptide-1) is a hormone involved in appetite and blood sugar regulation. Whenever you eat, your body releases a surge of GLP-1, which sends the “I’m full” signal to your brain. Orforglipron works by activating the same receptors as your body’s naturally occurring GLP-1, leading to decreased hunger and food intake (1).
Orforglipron is indicated for use in adults with a BMI of 30 or higher (obesity), or a BMI of 27 or greater (overweight) with at least one weight-related condition, to be used in conjunction with a reduced-calorie diet and increased physical activity (1). Orforglipron was approved by the FDA through the Commissioner’s National Priority Voucher pilot program, and was approved just 50 days after it was filed, making it the fastest new molecular entity approval since 2002 (1).
What Did the ATTAIN-1 Clinical Trial Show?
The ATTAIN-1 clinical trial demonstrated that orforglipron resulted in significant weight loss at 72 weeks compared to a placebo (2). The ATTAIN-1 trial was a phase 3, randomized double-blind study published in the New England Journal of Medicine which enrolled 3,127 adults with obesity (without diabetes), and compared the efficacy of three different doses of orforglipron (6mg, 12mg, 36mg) with a placebo (2). It's worth noting that the FDA-approved doses of orforglipron range from 0.8 mg to 17.2 mg (1).
The ATTAIN-1 trial showed the following average body weight reductions over 72 weeks across all randomized participants (2):
6 mg: 7.5% weight loss
12 mg: 8.4% weight loss
36 mg: 11.2% weight loss
Placebo: 2.1% weight loss
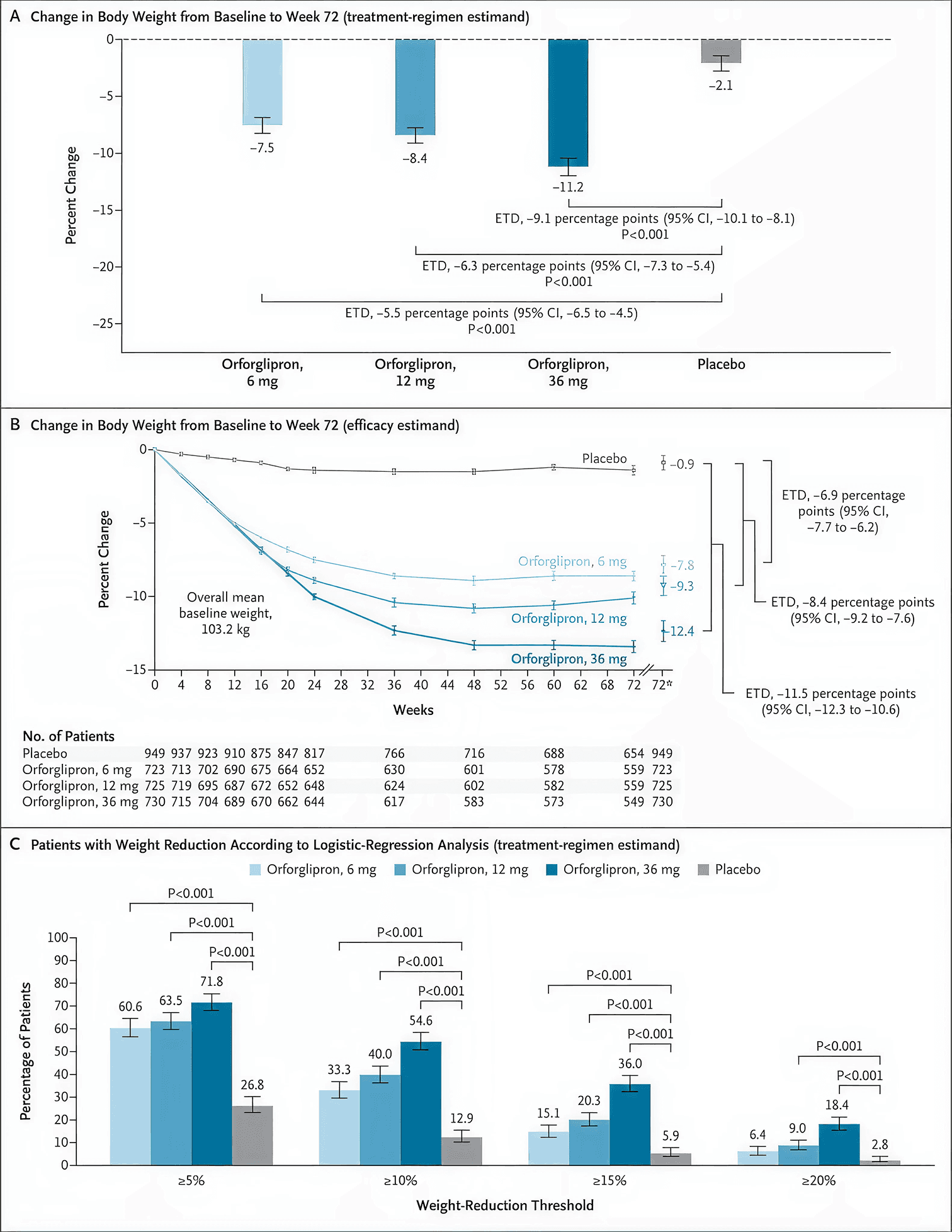
Figure 1. Weight loss results from the ATTAIN-1 trial by orforglipron dose (6 mg, 12 mg, and 36 mg) versus placebo over 72 weeks. Adapted from Wharton et al., NEJM 2025 (2).
Among participants who stayed on treatment for the full 72 weeks, the results were even more pronounced. The 12 mg group lost an average of 9.3% of their body weight, and the 6 mg group lost 7.8% (2). On the 12 mg dose, 63.5% of participants lost at least 5% of their body weight, and 40.0% lost at least 10% (2).
It wasn’t just the number on the scale that changed. The study also reported that people taking orforglipron had greater reductions in waist circumference, systolic blood pressure, triglycerides, and non-HDL cholesterol compared to those taking a placebo (2).
In total, two randomized, double-blind, placebo-controlled trials were used to support the FDA’s approval of orforglipron (1).
What Are the Side Effects of Orforglipron?
The majority of the side effects of orforglipron are gastrointestinal in nature, similar to other GLP-1 receptor agonists (1, 2). Common side effects listed by the FDA include (1):
Nausea
Constipation
Diarrhea
Vomiting
Indigestion (dyspepsia)
Stomach pain
Swollen belly (abdominal distension)
Fatigue
Belching
Heartburn (gastroesophageal reflux)
Gas
In the ATTAIN-1 trial, adverse events led to treatment discontinuation in 5.3% to 10.3% of people taking orforglipron, compared with 2.7% of people on placebo (2). The gastrointestinal side effects were mostly mild to moderate in severity (2).
Orforglipron also carries a boxed warning for thyroid C-cell tumors, including medullary thyroid carcinoma (1). It should not be used in anyone with a personal or family history of medullary thyroid cancer or Multiple Endocrine Neoplasia syndrome type 2 (1). Other serious warnings include pancreatitis, severe gastrointestinal reactions, acute kidney injury from volume depletion, low blood sugar (hypoglycemia), allergic reactions, gallbladder problems, diabetic retinopathy in people with type 2 diabetes, and pulmonary aspiration during general anesthesia or deep sedation (1).
If you notice side effects persisting or worsening, talk with your provider. Many GLP-1 side effects can be managed with dose adjustments and supportive care.
How Is Orforglipron Different From Other GLP-1 Medications?
Orforglipron is different from other injectable GLP-1 medications like injectable semaglutide and tirzepatide in several key ways. The most obvious is that it’s an oral medication, a pill you take by mouth rather than an injection, which could be a big plus if you’ve avoided medication because you don’t like needles (3).
But it’s not just about how you take it. Orforglipron is a small-molecule compound, not a peptide (2). Most current GLP-1 medications, including semaglutide and tirzepatide, are peptides, and that’s a big reason they have to be injected. Orforglipron is a nonpeptide compound that can survive digestion and is absorbed from a conventional tablet (2).
Convenience is also a factor. Convenience is a big part of what sets orforglipron apart from other oral GLP-1 weight loss options. Orforglipron is the only oral GLP-1 for weight loss that doesn't have to be taken at a specific time of day, and that has no restrictions on eating and drinking (3). If you've taken other oral GLP-1 medications, you know that they usually come with instructions on timing, fasting, and hydration. This flexibility may make it easier to incorporate orforglipron into your routine.
The flip side: orforglipron is a once-daily medication, while GLP-1 injectables like semaglutide and tirzepatide are usually injected once a week (3). So you'll trade the needle for a pill, but move from a weekly routine to a daily one.
How Much Does Orforglipron Cost?
Pricing is a major concern for anyone considering a GLP-1 medication, and as of April 2026, Eli Lilly has outlined several cost tiers for Foundayo (3):
Commercial insurance: Eligible patients may pay as little as $25 per month with the Foundayo savings card
Self-pay: Starting at $149 per month for the lowest dose
Medicare Part D: Eligible individuals may be able to access orforglipron for approximately $50 per month, beginning as soon as July 1, 2026
These prices are based on information from Eli Lilly at the time of FDA approval (3). Your costs may vary depending on your insurance, the pharmacy you use, and the dose your healthcare provider prescribes. Orforglipron is available in six doses: 0.8 mg, 2.5 mg, 5.5 mg, 9 mg, 14.5 mg, and 17.2 mg (3).
If cost has been a barrier to exploring weight loss treatment options, there are other options worth knowing about. On the Mochi Health telehealth platform, you can connect with a licensed healthcare provider who can prescribe either brand-name Foundayo or a compounded version of orforglipron, starting at $58.50 per month for the lowest dose. That may be a more affordable option for people without insurance coverage, or those who prefer a lower out-of-pocket expense.
Keep in mind that prices for GLP-1 medications may change over time, and it's a good idea to confirm current pricing with your provider or pharmacist.
Who Is Eligible for Orforglipron?
Orforglipron is approved by the FDA for use in adults with obesity (BMI 30 or above) or adults who are overweight (BMI 27 or above) with at least one weight-related condition (1). Orforglipron should be used in combination with a lower-calorie diet and increased exercise. It is not intended to be used alone for weight loss (1).
There are certain cases where you should not take orforglipron. According to the FDA and Eli Lilly, you should not take Foundayo if you (1, 3):
Have a personal or family history of medullary thyroid carcinoma
Have Multiple Endocrine Neoplasia syndrome type 2
Are currently taking another GLP-1 receptor agonist medication
Orforglipron has not been studied in children (3). It may also interfere with oral contraceptives, so Eli Lilly recommends using an alternative form of birth control for 30 days after starting orforglipron and for 30 days after each dose increase (3).
FAQ's
Is orforglipron the same as semaglutide or tirzepatide?
Although orforglipron, semaglutide, and tirzepitide are all GLP-1 receptor agonists, orforglipron is a small-molecule oral drug (2). Additionally, unlike other oral GLP-1 medications, it can be taken at any time of day without any food or water intake restrictions (3).
When will orforglipron be available at pharmacies?
Orforglipron (Foundayo) was FDA approved on April 1, 2026 (1). According to Eli Lilly, it began being available by prescription through LillyDirect immediately, and it will start shipping April 6, 2026 (3). It will also be available at retail pharmacies at a later date. You can speak with a licensed provider through Mochi Health’s telehealth platform to see if a prescription for Eli Lilly’s brand name Foundayo or a compounded version of orgorglipron is right for you.
Can I take orforglipron with my other medications?
You should not take orforglipron with another GLP-1 receptor agonist (1). Orforglipron may decrease the efficacy of oral contraceptives (3). Consult with your healthcare provider regarding all the medications you’re taking because some may need to be adjusted.
How much weight can I expect to lose on orforglipron?
Results will vary from person to person. In the ATTAIN-1 trial, participants on 12 mg dose lost an average of 8.4% of their body weight over 72 weeks, compared with 2.1% for placebo (2). Results may also depend on the dose your provider prescribes, your starting weight, and how consistently you follow a reduced-calorie diet and increased physical activity alongside the medication.
Does orforglipron cause nausea?
Nausea is the most common side effect of orforglipron, as with other GLP-1 drugs (1, 2). In the ATTAIN-1 study, gastrointestinal side effects were generally mild or moderate (2). Starting at a low dose and gradually increasing, as the dosing schedule is designed to do, may help manage these effects.
Check Your Eligibility
If you're interested in whether an oral GLP-1 medication like orforglipron could be a fit for your weight loss goals, a licensed provider on Mochi Health's telehealth platform can help you understand your options and determine the right path forward. Check your eligibility here.
Disclaimer: This article is for educational purposes only and should not be considered medical advice. The information provided does not constitute recommendations for treatment. Always consult with your healthcare provider about your specific situation, symptoms, and treatment options.
References
U.S. Food and Drug Administration. (2026, April 1). FDA approves first new molecular entity under national priority voucher program [Press release]. https://www.fda.gov/news-events/press-announcements/fda-approves-first-new-molecular-entity-under-national-priority-voucher-program
Wharton, S., Aronne, L. J., Stefanski, A., Alfaris, N. F., Ciudin, A., Yokote, K., Halpern, B., Shukla, A. P., Zhou, C., Macpherson, L., Allen, S. E., Ahmad, N. N., & Klise, S. R. (2025). Orforglipron, an oral small-molecule GLP-1 receptor agonist for obesity treatment. New England Journal of Medicine, 393(18), 1796-1806. https://doi.org/10.1056/NEJMoa2511774
Eli Lilly and Company. (2026, April 1). FDA approves Lilly's Foundayo (orforglipron), the only GLP-1 pill for weight loss that can be taken any time of day without food or water restrictions [Press release]. https://investor.lilly.com/news-releases/news-release-details/fda-approves-lillys-foundayotm-orforglipron-only-glp-1-pill
Key Takeaways
Orforglipron (brand name Foundayo) is a nonpeptide oral GLP-1 pill for weight loss, FDA-approved on April 1, 2026
Unlike injectable GLP-1 medications, orforglipron can be taken any time of day without food or water restrictions
In the ATTAIN-1 clinical trial, participants on 12 mg of orforglipron lost an average of 8.4% of their body weight over 72 weeks (the FDA-approved doses range up to 17.2 mg)
The most common side effects are gastrointestinal, including nausea, constipation, and diarrhea, and were mostly mild to moderate in clinical trials
Without insurance Foundayo will cost $149 per month at the lowest dose. At Mochi Health, you can also speak with an independent licensed provider about compounded orforglipron options, starting at $58.50 per month.
Orforglipron is a new FDA-approved oral weight loss medication, sold under the brand name Foundayo and manufactured by Eli Lilly (1, 3). It is a small molecule (non-peptide) GLP-1 receptor agonist which was approved on April 1, 2026, for use in adults with obesity or overweight with at least one weight-related condition (1). Unlike injectable GLP-1 receptor agonists, orforglipron is a once-daily oral medication that you can take at any time of day, with or without food, and without any water restrictions (3).
If you have been keeping up with the GLP-1 medications for weight loss, you will recognize how huge this is! In this article, we will cover: How orforglipron works, Results of the clinical trial, Side effects, How it compares to other GLP-1 receptor agonists, Cost of the medication, and Who may be eligable.
What Is Orforglipron and How Does It Work?
Orforglipron is a new FDA-approved oral weight loss medication, sold under the brand name Foundayo and manufactured by Eli Lilly (1, 3). It is a small molecule (non-peptide) GLP-1 receptor agonist which was approved on April 1, 2026, for use in adults with obesity or overweight with at least one weight-related condition (1). Unlike injectable GLP-1 receptor agonists like semaglutide or tirzepatide, orforglipron is a once-daily oral medication that you can take at any time of day, with or without food, and without any water restrictions (3).
GLP-1 (glucagon-like peptide-1) is a hormone involved in appetite and blood sugar regulation. Whenever you eat, your body releases a surge of GLP-1, which sends the “I’m full” signal to your brain. Orforglipron works by activating the same receptors as your body’s naturally occurring GLP-1, leading to decreased hunger and food intake (1).
Orforglipron is indicated for use in adults with a BMI of 30 or higher (obesity), or a BMI of 27 or greater (overweight) with at least one weight-related condition, to be used in conjunction with a reduced-calorie diet and increased physical activity (1). Orforglipron was approved by the FDA through the Commissioner’s National Priority Voucher pilot program, and was approved just 50 days after it was filed, making it the fastest new molecular entity approval since 2002 (1).
What Did the ATTAIN-1 Clinical Trial Show?
The ATTAIN-1 clinical trial demonstrated that orforglipron resulted in significant weight loss at 72 weeks compared to a placebo (2). The ATTAIN-1 trial was a phase 3, randomized double-blind study published in the New England Journal of Medicine which enrolled 3,127 adults with obesity (without diabetes), and compared the efficacy of three different doses of orforglipron (6mg, 12mg, 36mg) with a placebo (2). It's worth noting that the FDA-approved doses of orforglipron range from 0.8 mg to 17.2 mg (1).
The ATTAIN-1 trial showed the following average body weight reductions over 72 weeks across all randomized participants (2):
6 mg: 7.5% weight loss
12 mg: 8.4% weight loss
36 mg: 11.2% weight loss
Placebo: 2.1% weight loss

Figure 1. Weight loss results from the ATTAIN-1 trial by orforglipron dose (6 mg, 12 mg, and 36 mg) versus placebo over 72 weeks. Adapted from Wharton et al., NEJM 2025 (2).
Among participants who stayed on treatment for the full 72 weeks, the results were even more pronounced. The 12 mg group lost an average of 9.3% of their body weight, and the 6 mg group lost 7.8% (2). On the 12 mg dose, 63.5% of participants lost at least 5% of their body weight, and 40.0% lost at least 10% (2).
It wasn’t just the number on the scale that changed. The study also reported that people taking orforglipron had greater reductions in waist circumference, systolic blood pressure, triglycerides, and non-HDL cholesterol compared to those taking a placebo (2).
In total, two randomized, double-blind, placebo-controlled trials were used to support the FDA’s approval of orforglipron (1).
What Are the Side Effects of Orforglipron?
The majority of the side effects of orforglipron are gastrointestinal in nature, similar to other GLP-1 receptor agonists (1, 2). Common side effects listed by the FDA include (1):
Nausea
Constipation
Diarrhea
Vomiting
Indigestion (dyspepsia)
Stomach pain
Swollen belly (abdominal distension)
Fatigue
Belching
Heartburn (gastroesophageal reflux)
Gas
In the ATTAIN-1 trial, adverse events led to treatment discontinuation in 5.3% to 10.3% of people taking orforglipron, compared with 2.7% of people on placebo (2). The gastrointestinal side effects were mostly mild to moderate in severity (2).
Orforglipron also carries a boxed warning for thyroid C-cell tumors, including medullary thyroid carcinoma (1). It should not be used in anyone with a personal or family history of medullary thyroid cancer or Multiple Endocrine Neoplasia syndrome type 2 (1). Other serious warnings include pancreatitis, severe gastrointestinal reactions, acute kidney injury from volume depletion, low blood sugar (hypoglycemia), allergic reactions, gallbladder problems, diabetic retinopathy in people with type 2 diabetes, and pulmonary aspiration during general anesthesia or deep sedation (1).
If you notice side effects persisting or worsening, talk with your provider. Many GLP-1 side effects can be managed with dose adjustments and supportive care.
How Is Orforglipron Different From Other GLP-1 Medications?
Orforglipron is different from other injectable GLP-1 medications like injectable semaglutide and tirzepatide in several key ways. The most obvious is that it’s an oral medication, a pill you take by mouth rather than an injection, which could be a big plus if you’ve avoided medication because you don’t like needles (3).
But it’s not just about how you take it. Orforglipron is a small-molecule compound, not a peptide (2). Most current GLP-1 medications, including semaglutide and tirzepatide, are peptides, and that’s a big reason they have to be injected. Orforglipron is a nonpeptide compound that can survive digestion and is absorbed from a conventional tablet (2).
Convenience is also a factor. Convenience is a big part of what sets orforglipron apart from other oral GLP-1 weight loss options. Orforglipron is the only oral GLP-1 for weight loss that doesn't have to be taken at a specific time of day, and that has no restrictions on eating and drinking (3). If you've taken other oral GLP-1 medications, you know that they usually come with instructions on timing, fasting, and hydration. This flexibility may make it easier to incorporate orforglipron into your routine.
The flip side: orforglipron is a once-daily medication, while GLP-1 injectables like semaglutide and tirzepatide are usually injected once a week (3). So you'll trade the needle for a pill, but move from a weekly routine to a daily one.
How Much Does Orforglipron Cost?
Pricing is a major concern for anyone considering a GLP-1 medication, and as of April 2026, Eli Lilly has outlined several cost tiers for Foundayo (3):
Commercial insurance: Eligible patients may pay as little as $25 per month with the Foundayo savings card
Self-pay: Starting at $149 per month for the lowest dose
Medicare Part D: Eligible individuals may be able to access orforglipron for approximately $50 per month, beginning as soon as July 1, 2026
These prices are based on information from Eli Lilly at the time of FDA approval (3). Your costs may vary depending on your insurance, the pharmacy you use, and the dose your healthcare provider prescribes. Orforglipron is available in six doses: 0.8 mg, 2.5 mg, 5.5 mg, 9 mg, 14.5 mg, and 17.2 mg (3).
If cost has been a barrier to exploring weight loss treatment options, there are other options worth knowing about. On the Mochi Health telehealth platform, you can connect with a licensed healthcare provider who can prescribe either brand-name Foundayo or a compounded version of orforglipron, starting at $58.50 per month for the lowest dose. That may be a more affordable option for people without insurance coverage, or those who prefer a lower out-of-pocket expense.
Keep in mind that prices for GLP-1 medications may change over time, and it's a good idea to confirm current pricing with your provider or pharmacist.
Who Is Eligible for Orforglipron?
Orforglipron is approved by the FDA for use in adults with obesity (BMI 30 or above) or adults who are overweight (BMI 27 or above) with at least one weight-related condition (1). Orforglipron should be used in combination with a lower-calorie diet and increased exercise. It is not intended to be used alone for weight loss (1).
There are certain cases where you should not take orforglipron. According to the FDA and Eli Lilly, you should not take Foundayo if you (1, 3):
Have a personal or family history of medullary thyroid carcinoma
Have Multiple Endocrine Neoplasia syndrome type 2
Are currently taking another GLP-1 receptor agonist medication
Orforglipron has not been studied in children (3). It may also interfere with oral contraceptives, so Eli Lilly recommends using an alternative form of birth control for 30 days after starting orforglipron and for 30 days after each dose increase (3).
FAQ's
Is orforglipron the same as semaglutide or tirzepatide?
Although orforglipron, semaglutide, and tirzepitide are all GLP-1 receptor agonists, orforglipron is a small-molecule oral drug (2). Additionally, unlike other oral GLP-1 medications, it can be taken at any time of day without any food or water intake restrictions (3).
When will orforglipron be available at pharmacies?
Orforglipron (Foundayo) was FDA approved on April 1, 2026 (1). According to Eli Lilly, it began being available by prescription through LillyDirect immediately, and it will start shipping April 6, 2026 (3). It will also be available at retail pharmacies at a later date. You can speak with a licensed provider through Mochi Health’s telehealth platform to see if a prescription for Eli Lilly’s brand name Foundayo or a compounded version of orgorglipron is right for you.
Can I take orforglipron with my other medications?
You should not take orforglipron with another GLP-1 receptor agonist (1). Orforglipron may decrease the efficacy of oral contraceptives (3). Consult with your healthcare provider regarding all the medications you’re taking because some may need to be adjusted.
How much weight can I expect to lose on orforglipron?
Results will vary from person to person. In the ATTAIN-1 trial, participants on 12 mg dose lost an average of 8.4% of their body weight over 72 weeks, compared with 2.1% for placebo (2). Results may also depend on the dose your provider prescribes, your starting weight, and how consistently you follow a reduced-calorie diet and increased physical activity alongside the medication.
Does orforglipron cause nausea?
Nausea is the most common side effect of orforglipron, as with other GLP-1 drugs (1, 2). In the ATTAIN-1 study, gastrointestinal side effects were generally mild or moderate (2). Starting at a low dose and gradually increasing, as the dosing schedule is designed to do, may help manage these effects.
Check Your Eligibility
If you're interested in whether an oral GLP-1 medication like orforglipron could be a fit for your weight loss goals, a licensed provider on Mochi Health's telehealth platform can help you understand your options and determine the right path forward. Check your eligibility here.
Disclaimer: This article is for educational purposes only and should not be considered medical advice. The information provided does not constitute recommendations for treatment. Always consult with your healthcare provider about your specific situation, symptoms, and treatment options.
References
U.S. Food and Drug Administration. (2026, April 1). FDA approves first new molecular entity under national priority voucher program [Press release]. https://www.fda.gov/news-events/press-announcements/fda-approves-first-new-molecular-entity-under-national-priority-voucher-program
Wharton, S., Aronne, L. J., Stefanski, A., Alfaris, N. F., Ciudin, A., Yokote, K., Halpern, B., Shukla, A. P., Zhou, C., Macpherson, L., Allen, S. E., Ahmad, N. N., & Klise, S. R. (2025). Orforglipron, an oral small-molecule GLP-1 receptor agonist for obesity treatment. New England Journal of Medicine, 393(18), 1796-1806. https://doi.org/10.1056/NEJMoa2511774
Eli Lilly and Company. (2026, April 1). FDA approves Lilly's Foundayo (orforglipron), the only GLP-1 pill for weight loss that can be taken any time of day without food or water restrictions [Press release]. https://investor.lilly.com/news-releases/news-release-details/fda-approves-lillys-foundayotm-orforglipron-only-glp-1-pill
Read next
Ready to transform your health?
Unlock access to expert guidance and a weight care plan crafted just for you.

© 2026 Mochi Health
All professional medical services are provided by licensed physicians and clinicians affiliated with independently owned and operated professional practices. Mochi Health Corp. provides administrative and technology services to affiliated medical practices it supports, and does not provide any professional medical services itself.


© 2026 Mochi Health
All professional medical services are provided by licensed physicians and clinicians affiliated with independently owned and operated professional practices. Mochi Health Corp. provides administrative and technology services to affiliated medical practices it supports, and does not provide any professional medical services itself.


© 2026 Mochi Health
All professional medical services are provided by licensed physicians and clinicians affiliated with independently owned and operated professional practices. Mochi Health Corp. provides administrative and technology services to affiliated medical practices it supports, and does not provide any professional medical services itself.